
Marcia Bergeron died of
metal toxicity caused by counterfeit drugs she bought
online
Photo: Handout photo |
At first, BC Coroners Service case
number 2006:0143:0117 was something of a mystery. The
case involved Quadra Island resident Marcia Bergeron,
58, who was found dead in her bed by a friend last December.
"We had no reasonable cause of
death, and she was of an age where sudden death might
not be expected," says Vancouver Island regional coroner
Rose Stanton, who worked on the file. "There was no
evidence of suicide, foul play or trauma. But there
were certainly some medications we could not explain
or identify." Unmarked packages and bags of pills were
found, which turned out to contain the hypnotic zolpidem,
which is not available in Canada, the prescription anti-anxiety
drug alprazolam and acetaminophen. Coroners Service
policy dictates that when drugs are present in an unexplained
death, a toxicology examination be conducted. Ms Stanton
received the toxicology report in April. "It was quite
extensive, as it turned out," she says.
That's putting it mildly. The Burnaby,
BC, lab found diphenhydramine (an OTC sedative), acetaminophen,
mirtzapine (a prescription antidepressant), dextromethorphan
(an OTC cough medicine) and methocarbamol (an OTC muscle
relaxant). But most shocking were the dangerously high
levels of metals, including aluminum (15 µg/g),
phosphorus, titanium, tin, strontium, arsenic and other
metals in Ms Bergeron's liver. The level of aluminum
alone was 15 times higher than what's considered normal.
So how did Marcia Bergeron end
up with a heady cocktail of drugs and metals in her
system? More importantly, did they kill her?
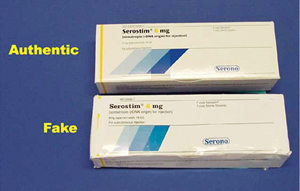
FDA comparison of real
and counterfeit Serostim (somatrapin), a human growth
hormone used to treat HIV wasting
Photo: FDA |
ELECTRONIC
CLUES
RCMP and US Food and Drug Administration
(FDA) officials cracked the case when they took a look
at Ms Bergeron's computer in March. She had been visiting
websites that sold unlicensed pharmaceuticals —
the kind you often see advertised in spam emails. Sifting
through her email account, investigators found a series
of messages to a friend in which she detailed side effects
she was suffering: nausea, diarrhea, aching joints,
and eventually hair loss, exhaustion and vision problems.
All are consistent with the meds that showed up on her
tox screen.
The electronic clues clinched it.
The drugs Ms Bergeron bought online were counterfeits;
they were manufactured poorly and contained impure "filler"
materials. It's since become apparent that the filler
was likely contaminated by dangerous metals during production
in Southeast Asia and sold by a website based in Eastern
Europe, possibly the Czech Republic, says Ms Stanton.
With all the pieces of the puzzle
finally in place, the BC Coroners Service inquiry wrapped
up on July 4. Medical cause of death: cardiac arrhythmia
caused by metal toxicity. By what means: ingestion of
contaminated counterfeit medication. Classification:
accidental.
PUBLIC
HAZARD
Ms Bergeron is the first person known to die as a result
of ingesting counterfeit meds. For physicians and regulators
her sad case is proof positive that drug counterfeiting
is a major public health hazard.
"People have been waiting for the
canary in the mineshaft," says Ms Stanton. Now they've
got one. With Marcia Bergeron, the FDA has finally been
able to directly link counterfeiting online pharmacies
to a patient's death. Prior cases had all lacked definitive
evidence connecting a fatal medication to a website.
Despite the paucity of direct links,
it's widely accepted that pharmaceutical counterfeiting
is increasing and that there are more ways now than
ever before to procure drugs from unlicensed sources.
The WHO estimates that between 1% and 10% of drugs sold
around the world are counterfeits, and as many as 50%
in some countries.
As demonstrated by the Bergeron
case, the problem exists in Canada as well. "It's something
that everyone has become more aware of," says David
W Johnston, president of the Canadian Association for
Pharmacy Distribution Management (CAPDM). A 2005 FDA
sting operation in New York, Miami and LA airports discovered
that almost 50% of drugs shipped to US customers from
four selected countries claimed to be from "Canadian
pharmacies." In fact, just 15% of them were; the rest
came from any of 27 other countries.
CANADA'S
RESPONSE
Is Health Canada doing enough to protect patients from
the dangers of drug counterfeiting and internet pharmacies
that besmirch Canada's good name? "I would say they
are almost certainly not," says Dr James Wright, a UBC
professor of internal medicine and pharmacology and
managing director of the Therapeutics Initiative with
an interest in internet pharmacy trends and counterfeiting.
"There is almost no precedent, and there is really very
little control over it."
In March, the Canadian International
Pharmacy Association said Health Canada should rein
in those selling fake medication.
But Health Canada spokesperson
Alastair Sinclair says the government's Health Products
and Food Branch (HPFB) Anti-Counterfeit Strategy for
health products is nearly done and will be published
in the fall. Health Canada officials have already begun
looking at possible changes in legislation, including
the federal Food and Drugs Act, to strengthen the government's
ability to combat drug counterfeiting. In March, the
HPFB sent recommendations to Canadian pharmacists to
help them identify counterfeit products, both by ID
numbers and visual inspection.
Until the government gets a handle
on the issue, says Dr Wright, physicians must be more
vigilant. "We are not very good at asking patients what
medications they are taking other than what we are prescribing,"
he says.
For more on medication safety,
see "Help patients help themselves with prescription
drugs" on page 11.
|